Abstract
Context:
Chronic myelomonocytic leukemia (CMML) is a myeloid hematological malignancy close to myelodysplastic syndromes (MDS) (Itzykson, Leukemia 2013).
The role of tumor microenvironment (TME) in the pathogenesis of MDS is increasingly highlighted, particularly the interactions between mesenchymal stem cells (MSCs) and hematopoietic tumor cells within the endosteal niche (Raaijmakers, Nature 2010). Studies about CMML TME focused on the vascular niche and demonstrated an increased angiogenesis and higher medullar vascular endothelial growth factor rates (Aguayo, Blood 2000; Bellamy, Blood 2001). MSCs have been less studied in the specific subset of CMML.
Moreover, cancer is associated with a procoagulant state participating in tumor development and metastasis process (d'Asti, Meth Mol Biol 2016). Monocytes are important cells regarding coagulation as they shed procoagulant, tissue factor (TF)-bearing microparticles (del Conde, Blood 2005).
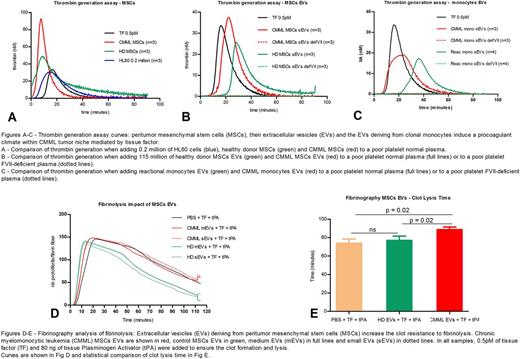
We hypothesized that MSCs and clonal monocytes induce a procoagulant "climate" within the tumor endosteal niche in CMML patients, via the extracellular vesicles (EVs) they release. This procoagulant state could participate to tumor development.
Methods:
We isolated MSCs from CMML (n=3) and control (n=3) patients' marrow aspirations, monocytes from CMML (n=3) and reactional monocytosis (n=4) patients' blood. We also cultured HL60 cell line as a reference, as HL60-derived EVs are procoagulant (Gheldof, Thromb Res 2014).
HL60 and MSCs were cultured with classical conditions and incubated in a medium free of serum EVs 72 hours before EVs collect. Monocytes were cultured for 40 hours in a serum-free medium to induce vesiculation and prevent contamination from serum EVs.
EVs were extracted by sequential centrifugations of culture supernatant. The pellets obtained after 10 000 g and 100 000 g ultra-centrifugations corresponded to the « medium EVs » containing mostly microparticles and « small EVs » respectively, containing exosomes along with small microparticles (Tkach, Cell 2016).
Concentration, mean size and size distribution of these EVs were analyzed by Nanoparticle Tracking Analysis (NS300, Malvern, UK). We tested always the same number of EVs for hemostasis tests.
The exploration of coagulation was mainly performed with thrombin generation assay (TGA). Phospholipids were systematically added so that the phospholipid concentration would not limit the coagulation reaction.
A fibrinolysis assay was performed on EVs deriving from MSCs and HL60 with a new technique currently developed: fibrinography. Relying on fibrin properties of light diffusion, it is a structural analysis of the nanostructure of the fibrin clot displaying the number of protofibrils per fibrin fiber during the coagulation and lysis reactions.
Results:
We showed that the vesiculation rate of MSCs (i.e. the number of EVs released by one cell) is significantly higher than the vesiculation rate of HL60 cells and monocytes (p = 0.02). The mean size of medium EVs (226 nm) was significantly higher than the mean size of small EVs (188 nm, p = 0.01).
TGA data are shown in the figure 1. With poor-platelet normal plasma, we showed that CMML MSCs but not control MSCs have a higher procoagulant activity than HL60 cells and 0.5pM of TF (Fig. 1A). We also demonstrated that small EVs derived from CMML monocytes and MSCs, but not from control cells, display a procoagulant activity (Fig. 1B and 1C).
TGA experiments were repeated with FVII-deficient plasma and did not display any thrombin generation when EVs were added. This implies that EVs procoagulant activity is mediated by TF present in the EVs.
Fibrinography coagulation data confirmed the TGA results and displayed a higher sensitivity than TGA during the early phase of coagulation. Analysis of fibrinolysis data showed a longer clot lysis time in presence of CMML small or medium EVs (p = 0.02) compared to healthy controls, corresponding to a higher clot resistance to fibrinolysis (Fig. 2).
Conclusion:
Overall, our findings reveal a procoagulant "climate" within the endosteal tumor niche in CMML mediated by clonal monocytes but also TME. This may induce a net of fibrin in the tumor hematopoietic niche, which could participate in leukemia progression.
Park: Novartis: Membership on an entity's Board of Directors or advisory committees, Research Funding; Hospira/Pfizer: Research Funding; Celgene: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal